ESG Integration of ‘Access to Medicine’ for the Pharmaceutical Sector
Reported by Franz Hong(Data Scientist/ESG Analyst)and Ingo Tietböehl(Director of ESG Research and Solutions)
Highlights:
・The Access to Medicine Index(AMI) can help forecast financial and stock performance of pharmaceutical companies by providing insight into their performance related to access to medicine in low- and middle-income countries.
・The Antimicrobial Resistance Benchmark (AMR) score itself, and especially the ‘manufacturing’ subscore are correlated with ROE & ROA, as they reflect pharmaceutical companies’ operational performance.
Correlations between AMR scores and ESG ratings are rather limited.・The top-performing companies in the 2022 AMI were GlaxoSmithKline, Johnson & Johnson and AstraZeneca. Japanese companies are lagging with Takeda as the only company in the top 10.
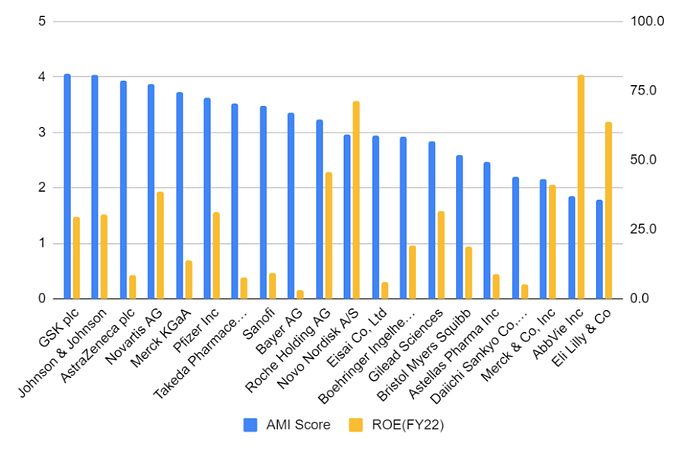
Figure 1: AMI score (2022) and ROE (2022) of top 20 companies
Introduction In summary, the Access to Medicine Index provides a comprehensive evaluation of pharmaceutical companies’ efforts to improve access to medicine in low- and middle-income countries. The index helps ESG analysts identify companies that are leaders in this area and may be better positioned for long-term success. It also helps identify potential risks and opportunities related to access to medicine and can be used to evaluate the alignment between a company’s values and objectives and those of its stakeholders. The top-performing companies in the 2022 index were GlaxoSmithKline, Johnson & Johnson and AstraZeneca.
The Access to Medicine Index is a valuable tool for evaluating the performance of pharmaceutical companies in improving access to medicine in low- and middle-income countries. The index helps to identify companies that are likely to perform well in the long term by evaluating companies’ policies, practices, and performance related to access to medicine. Companies that are leaders in this area may be better positioned to meet the needs of patients in low- and middle-income countries and may be more resilient in the face of changing market conditions and regulatory environments.
The index also helps to identify potential risks and opportunities. Companies that are lagging behind in this area may face reputational and regulatory risks, while companies that are innovating and leading the way may have a competitive advantage in the market..
The Access to Medicine Index is an independent ranking of pharmaceutical companies that measures their efforts to improve access to medicine in low- and middle-income countries. The index was launched in 2008 by the Access to Medicine Foundation (AMF), a non-profit organisation based in the Netherlands. The index is updated every two years, and each edition includes a ranking of the top 20 pharmaceutical companies based on their performance. Meanwhile, AMF also publishes additional assessments, for example, Index Disease Focus (IDF) in 2010, and the Antimicrobial Resistance Benchmark (AMR) in 2021.
The index assesses companies’ policies, practices, and performance in multiple areas, these include: research and development, pricing and registration, and manufacturing and distribution. The criteria generally cover aspects such as governance of access, research and development, pricing, manufacturing and product delivery, public policy, market influence and compliance, capacity building, patents and licensing, and product donations.
As a means to urge healthcare companies to make a change, AMI provides investors with insights as to what companies are good candidates for portfolio, and how to help investee companies improve. According to Access to Medicine Foundation, more than 130 institutional investors, accounting for a total of US$ 21 trillion in assets under management, are using their research, including AMI, to make better informed investment decisions.
Widely recognised ESG rating agencies such as MSCI, S&P etc., also include access to healthcare as one of the criteria in their ESG ratings of pharmaceutical companies. Recognising the importance of healthcare access for the general population, MSCI also has a World Health Care Index focused on the healthcare sector.
This report analyses the relations between AMI/AMR and certain financial indicators, as well as ESG performance (MSCI ESG ratings, S&P ESG ratings).
AMI/AMR Score Methodology
The Access to Medicine Index is an independent ranking of pharmaceutical companies that measures their efforts to improve access to medicine in low- and middle-income countries. The index was launched in 2008 by the Access to Medicine Foundation, a non-profit organisation based in the Netherlands. The Access to Medicine Index is updated every two years, and each edition includes a ranking of the top 20 pharmaceutical companies based on their performance.
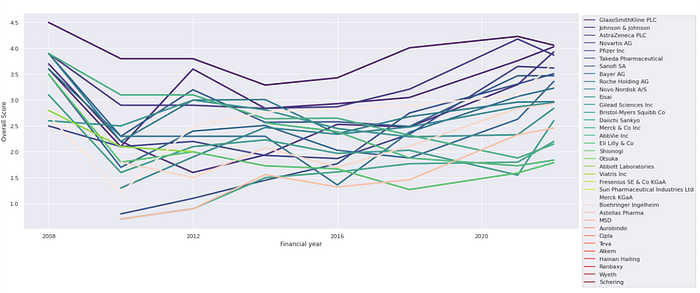
Figure 2 shows the overall score trend of all companies covered by AMI. Some companies consistently perform better than others, for example, GlaxoSmithKline PLC, Novartis AG, and Johnson & Johnson. On the other hand, Merck & Co Inc, Eli Lilly & Co, and AbbVie Inc saw general declining trend, while Japanese companies Takeda, Daiichi Sankyo, Eisai, and others like Pfizer Inc, Astellas Pharma, Boehringer Ingelheim improved gradually.
Figure 2: Overall score trend of analysed companies (Click here for an interactive graph)
The purpose of the Access to Medicine Index is to encourage pharmaceutical companies to prioritise access to medicine in their business strategies and to provide a tool for investors, governments, and other stakeholders to evaluate their performance in this area. The index is based on a set of 33 indicators that assess companies’ policies, practices, and performance in three areas: research and development, pricing and registration, and manufacturing and distribution.
The company list and criteria being evaluated changed slightly over the years, nevertheless, the criteria generally cover the following aspects, namely, a) Governance of Access, b) Research & Development, c) Pricing, Manufacturing & Product Delivery, d) Public Policy, Market Influence & Compliance, e) Capacity Building, f) Patents & Licensing, and g) Product Donations.
Governance of Access: Companies are assessed on the basis of their commitment to access to medicine, their policies and management systems related to access to medicine, and their engagement with stakeholders.
Research and Development: Companies are evaluated on the basis of their efforts to develop products for diseases that disproportionately affect low- and middle-income countries, their collaboration with other stakeholders, and their strategies for sharing knowledge and intellectual property.
Pricing: Companies are assessed on the basis of their pricing strategies and their efforts to make medicines affordable and accessible in low- and middle-income countries, including through voluntary licensing and pricing agreements.
Manufacturing: Practices of pharmaceutical companies by evaluating their efforts to ensure the quality, safety, and efficiency of their manufacturing processes. The index looks at how companies manage their supply chains, including their sourcing of raw materials and their relationships with suppliers, and evaluates the extent to which companies apply Good Manufacturing Practices (GMP) in their facilities.
Product Distribution/Delivery: Companies are evaluated on the basis of their efforts to ensure that their products reach patients in low- and middle-income countries, including through partnerships with local healthcare providers and distributors.
Public Policy, Market Influence & Compliance: examines how companies self-regulate their behaviour, and reports on whether companies have been found in breach of relevant laws and standards in all countries in which they operate. The Index views breaches wherever they occur as an indication of whether company-wide policies and codes are functioning.
Capacity Building: Companies are assessed on the basis of their efforts to build healthcare capacity in low- and middle-income countries, including through training and education programs for healthcare providers and support for healthcare infrastructure.
Patents & Licensing: Companies are analysed based on their management and efforts regarding IP as means to enable access to affordable medicines where they are needed, to encourage the entry of generic medicine manufacturers, and to stimulate market creation.
Product donations: looks at the scale of donation programmes in terms of the number of people reached, the financial value, and the number of disease-endemic countries in which the donation programme is active. This aspect also measures whether companies monitor the outcomes and impact of donation programmes, and try to maximise its effectiveness, and the processes companies have in place to ensure they can respond rapidly to emergency situations or humanitarian crises.
Analysis: Correlation between AMI/AMR, financial performance and ESG rating
Summary: AMI/AMR score is a leading indicator for financial performance, especially PBR. However, correlation with ESG performance/ESG ratings is limited.
As figure 3 highlights that AMI score itself is highly correlated with ROE, ROA and PBR in FY+2. However, in order to forecast financial performance, ‘Patents & Licensing’ score seems to be most suitable.
Figure 3. Correlation results between AMI/AMR scores and financial performance lagging two years

Regarding the correlation between AMI/AMR and ESG scores, Surprisingly, AMI/AMR does not show clear correlation with MSCI or S&P ESG ratings. AMR’s manufacturing is moderately correlated with MSCI ESG ratings, indicating that other manufacturing related elements play a greater role in the MSCI ESG rating. This is now major surprise given that ‘Manufacturing’ includes many criteria that determine the operational effectiveness and efficiency of pharmaceutical companies’ global manufacturing processes, thus reflecting operational performance. In fact “Product Safety & Quality” makes up around 27% of pharmaceuticals’ ESG score, while “Access to Health Care” constitutes for only 12%.
Additionally, AMI’s Research & Development has high correlation with S&P ESG ratings in the current year and two years later. This highlights the importance of R&D in the S&P rating, in which “Customer engagement” accounts for 35% of the social score. “Customer engagement” includes metrics such as “Spending on R&D as a percentage of revenue” and “Percentage of revenue from products launched in the past 3 years (branded drugs)”, which are closely connected to R&D efforts & pipeline of pharmaceutical companies.
The difference in correlation between AMR/AMI and MSCI/S&P ESG ratings highlights again that materiality and metrics weights are still very different among rating vendors.
Future prospects The Access to Medicine Index has the potential to drive greater accountability and transparency in the pharmaceutical industry by continuing to evaluate companies’ efforts to improve access to medicine in low- and middle-income countries. As the index evolves, it may incorporate new criteria and indicators that reflect emerging issues and trends in the pharmaceutical industry, such as the development of new therapies for neglected diseases or the adoption of sustainable manufacturing practices.
In addition, the Access to Medicine Index can play a key role in promoting greater collaboration and knowledge-sharing among pharmaceutical companies, governments, and other stakeholders. By highlighting best practices and identifying areas for improvement, the index can encourage companies to learn from one another and work together to address common challenges in global healthcare.
Moreover, the Access to Medicine Index may help promote more meaningful engagement between investors and pharmaceutical companies on issues related to access to medicine. By providing a common framework for evaluating companies’ performance in this area, the index can facilitate more informed and constructive dialogue between investors and companies, which can ultimately lead to improved performance and greater social impact.
However, the Access to Medicine Index may face challenges in keeping pace with the rapidly evolving pharmaceutical industry and the complex global health landscape. The index will need to continue to evolve and adapt to changing conditions, while maintaining its independence and credibility as an objective evaluator of pharmaceutical companies’ performance in improving access to medicine.
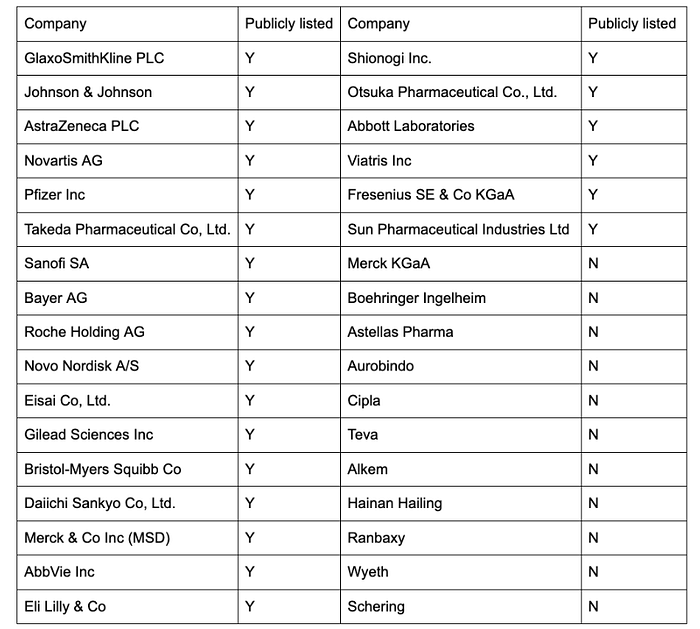
Figure 4 . Companies assessed in AMI (2008–2022)